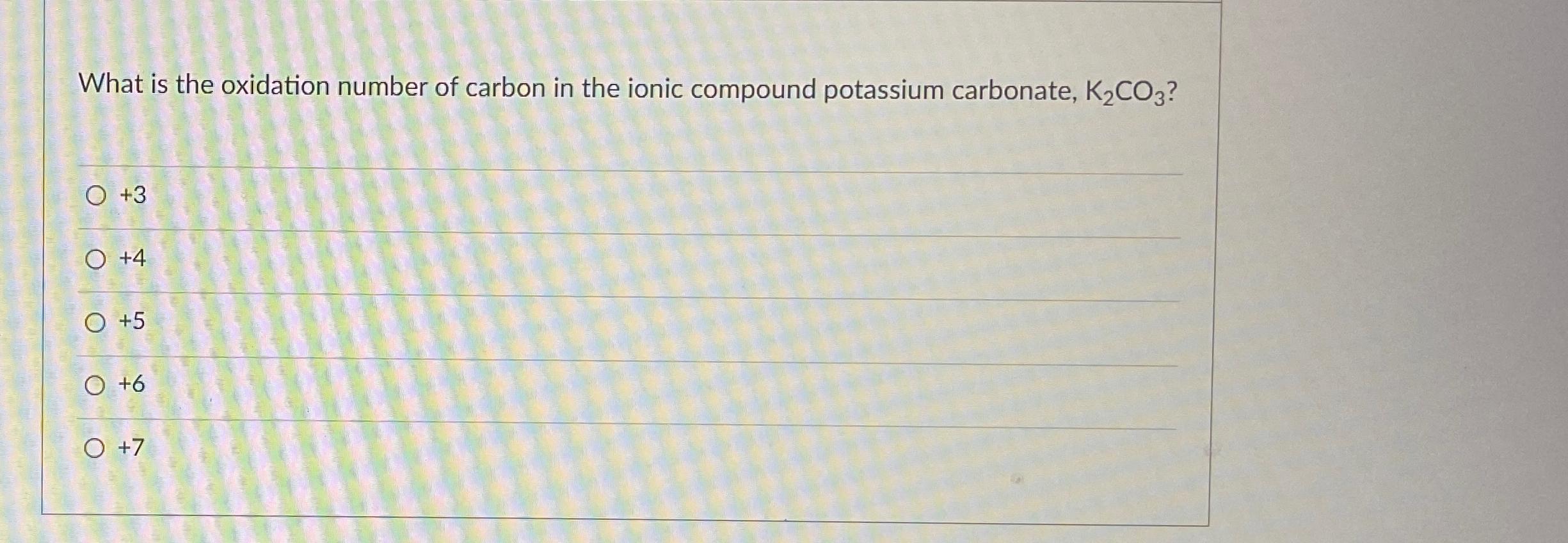
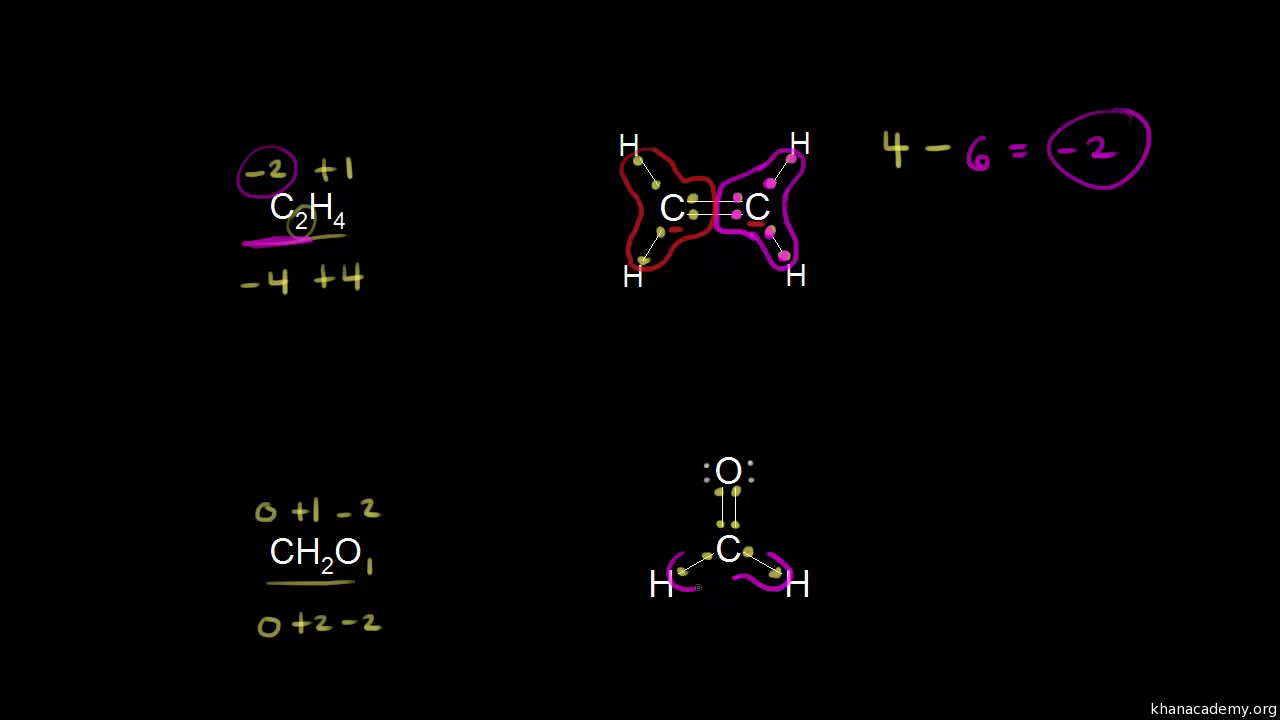
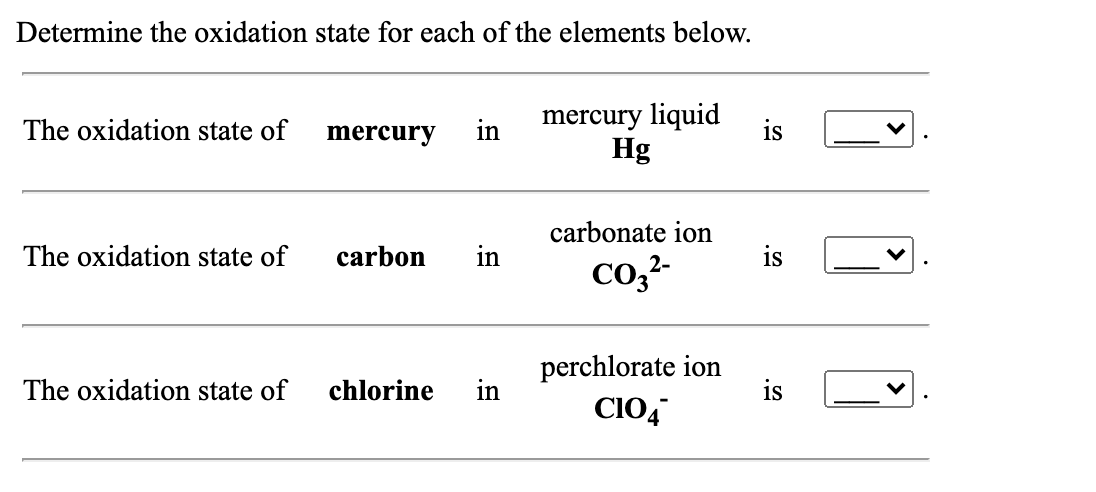
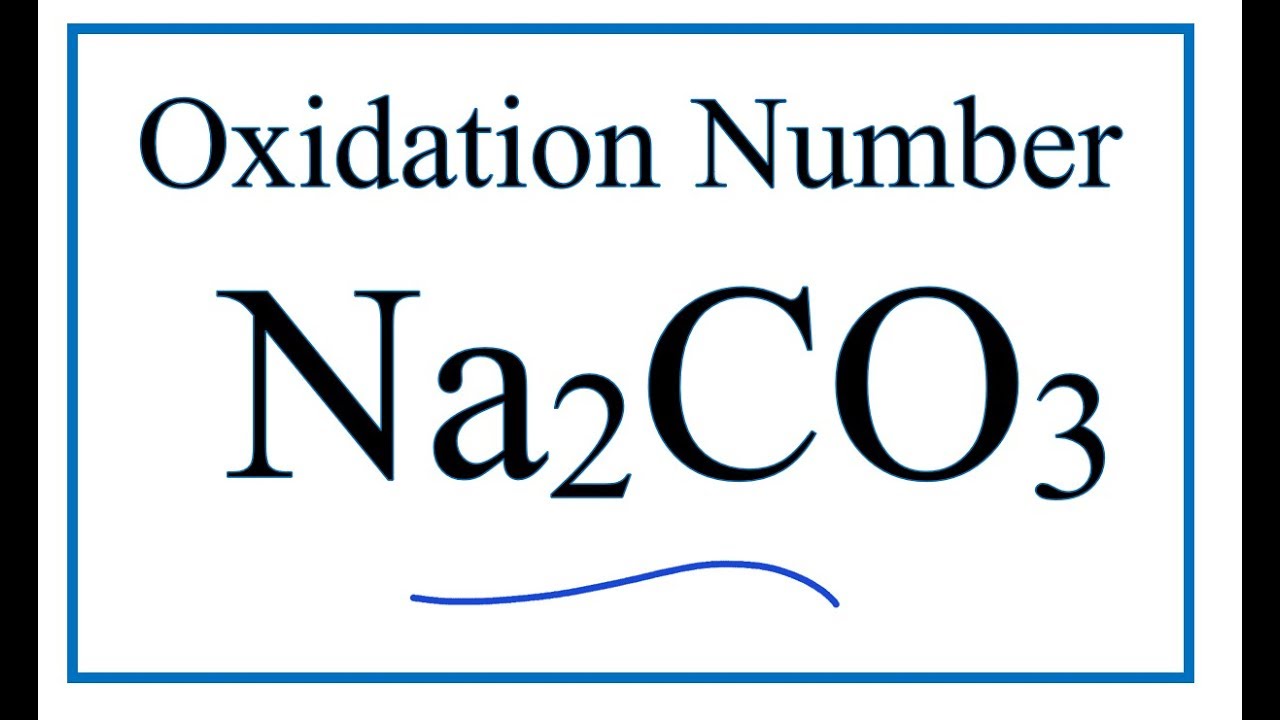SOLVED: The oxidation number of the carbon atom in cesium oxalate, Cs2C2O4, is: A +1 +6 +2 -2 -8 B +2 C +3 D +6. 2500 mL of carbonate ion is titrated with.

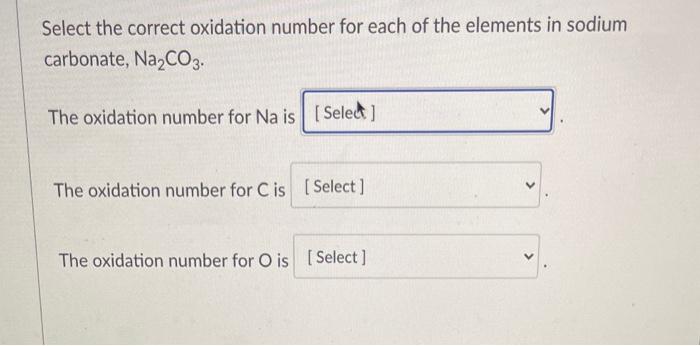
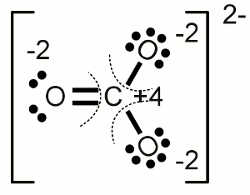
What is oxidation state of carbon in manganese carbonate MnCO3 - Chemistry - Redox Reactions - 12734691 | Meritnation.com




![ANSWERED] What is the oxidation number for carbon in the ionic - Kunduz ANSWERED] What is the oxidation number for carbon in the ionic - Kunduz](https://media.kunduz.com/media/sug-question/raw/80184035-1659893174.8312.jpeg)