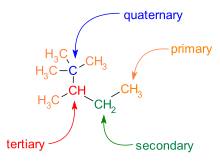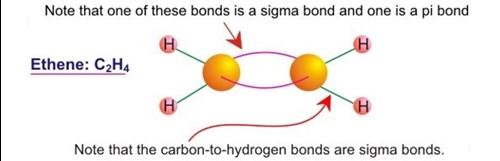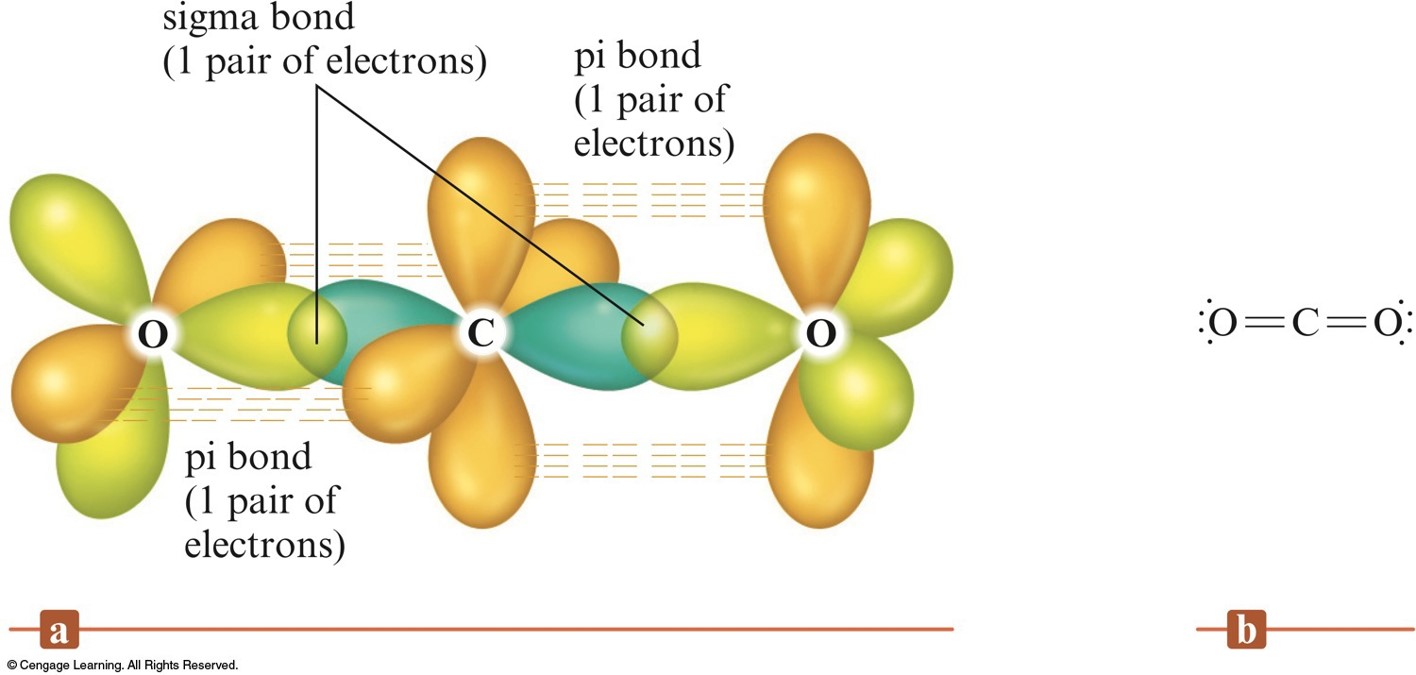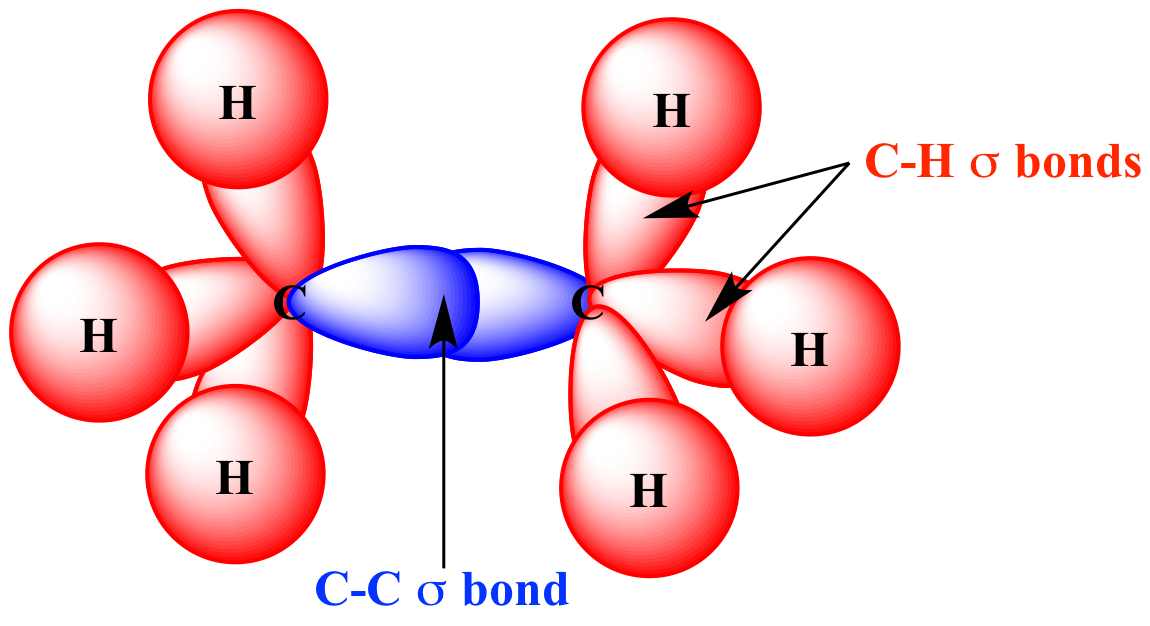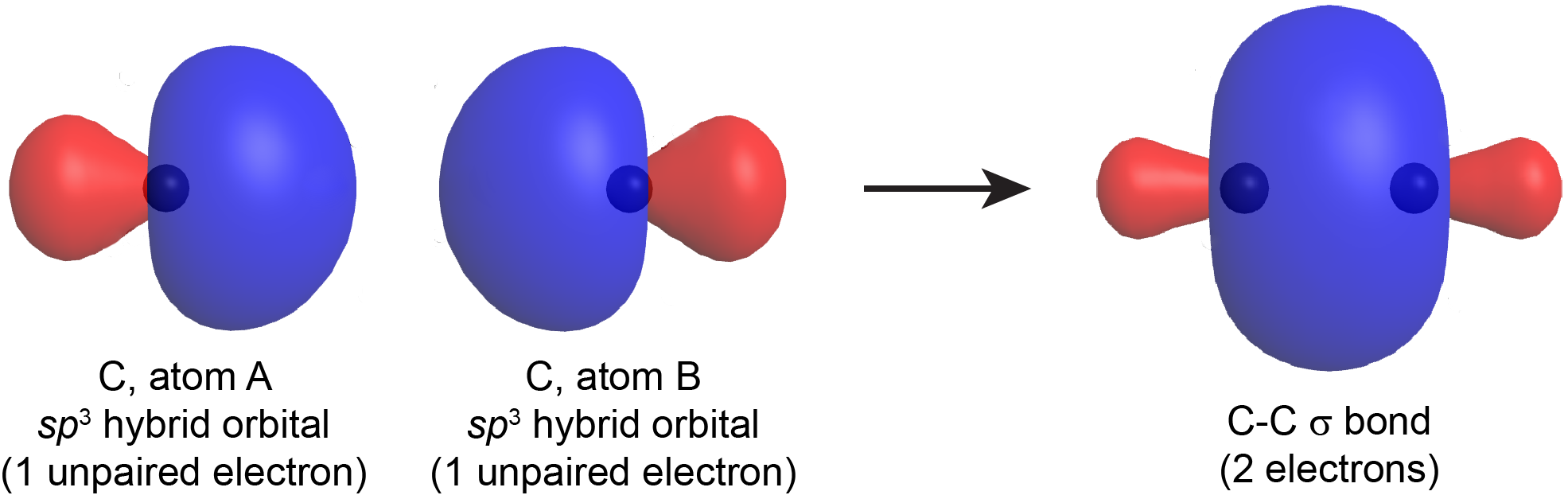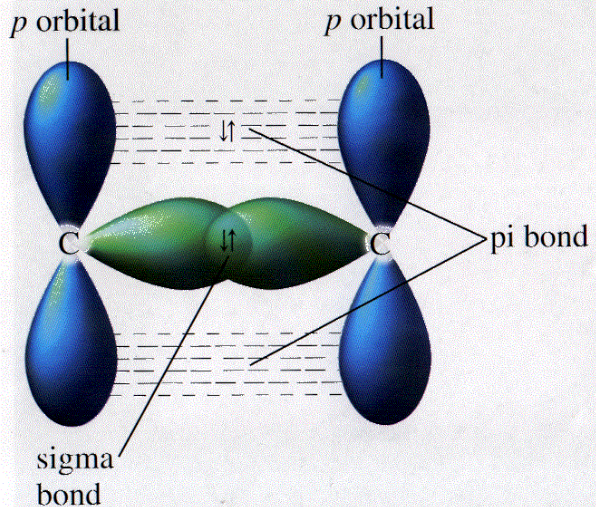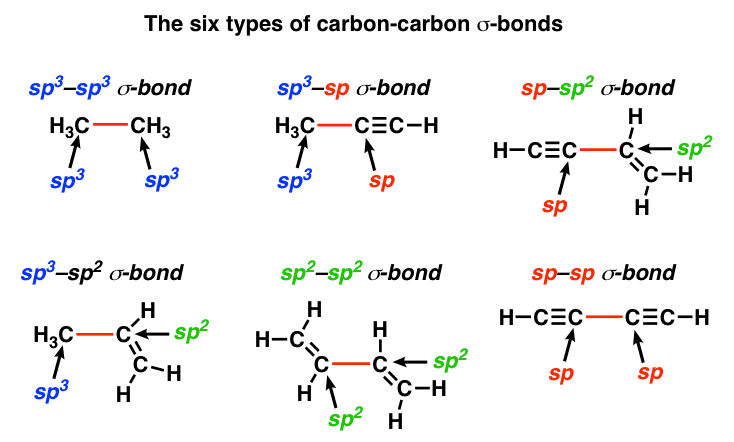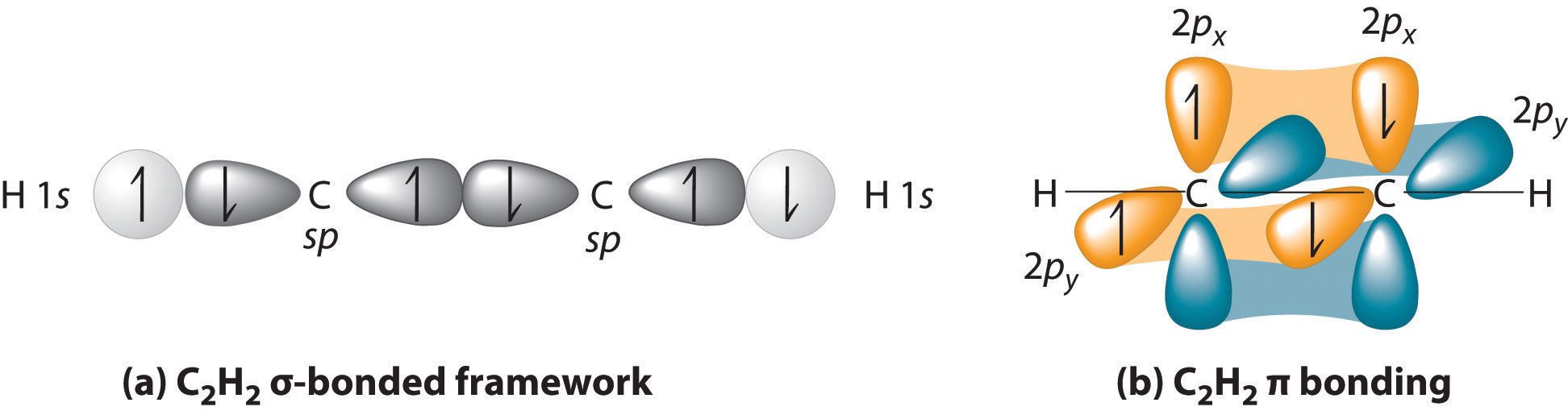
How does carbon use its "s" and "p" orbitals to form bonds in ethyne, ethene, and ethane? | Socratic

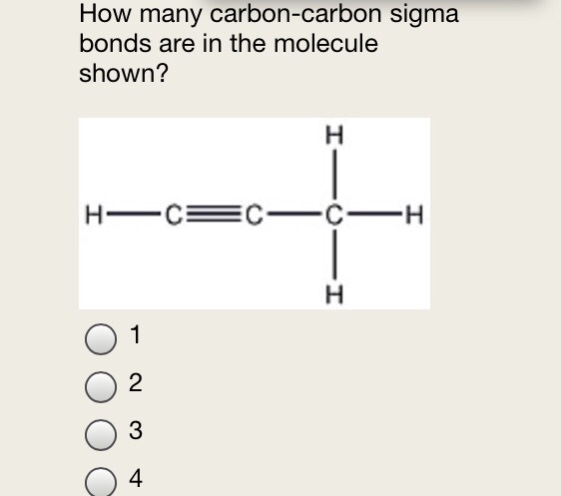
How many carbon-carbon Sigma bonds are in the molecule shown? a) 1 b) 2 c) 3 d) 4 | Homework.Study.com

Using diagrams, explain the difference between the and the bond in the carbon-carbon double bond of an alkene. | Homework.Study.com
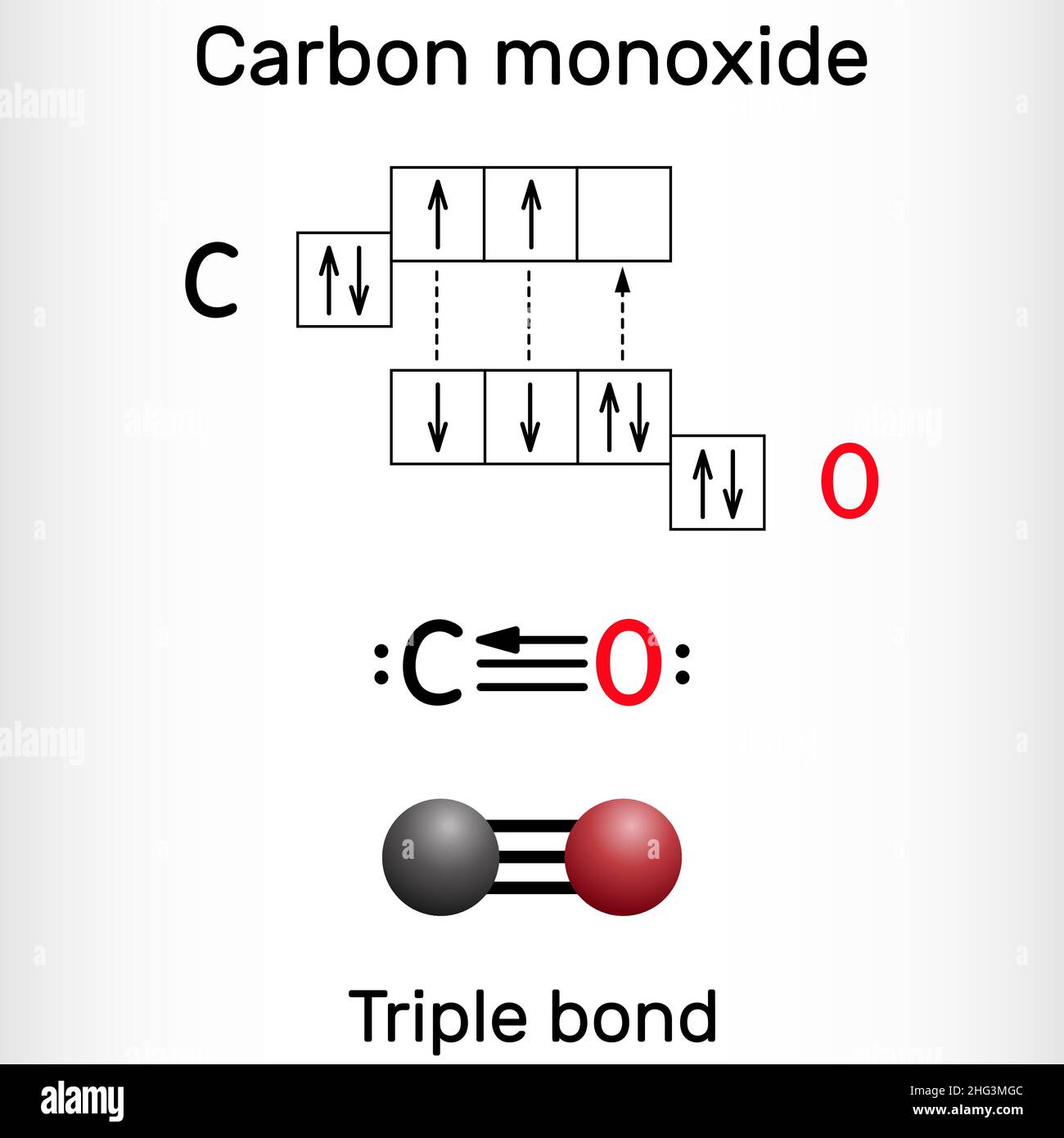

/chapter1/pages13and14/page13and14_files/chmo.png)