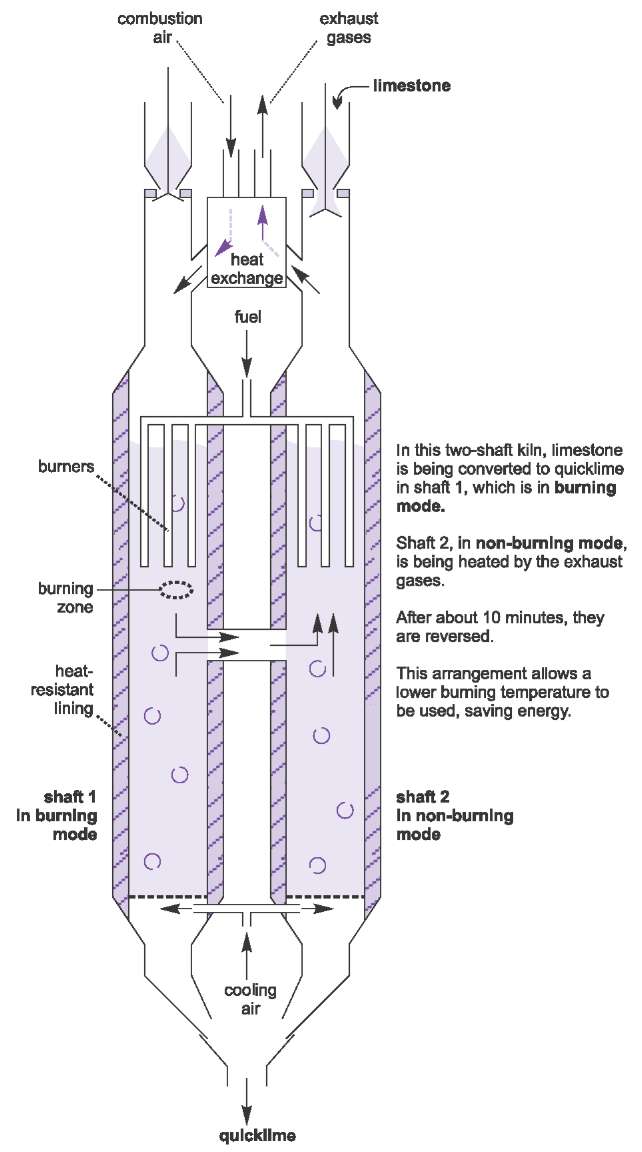
Reductive calcination of calcium carbonate in hydrogen and methane: A thermodynamic analysis on different reaction routes and evaluation of carbon dioxide mitigation potential - ScienceDirect
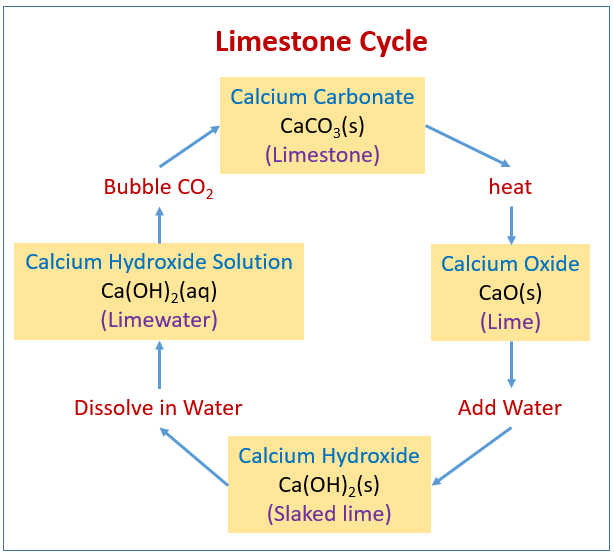
![Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f](https://c8.alamy.com/comp/2CJ846R/principles-of-agricultural-chemistry-microform-sea-water-is-decomposedsetting-carbon-dioxide-free-and-the-calcium-carbonate-is-used-bythe-animal-to-form-its-shell-cauco-=-cacog-hoo-f-cothis-carbon-dioxide-was-originally-derived-from-the-air-5-dissociation-of-carbonates-by-heat-as-in-the-burning-oflime-this-is-a-matter-of-small-importance-especially-as-thelime-takes-up-the-carbon-dioxide-again-sooner-or-later-cacos-=-cao-co-carbon-dioxide-is-also-emitted-from-some-volcanoes-deepsprings-and-other-subterranean-sources-quantity-presentcountry-air-contains-on-an-av-2CJ846R.jpg)
Principles of agricultural chemistry [microform] . sea water is decomposed,setting carbon dioxide free, and the calcium carbonate is used bythe animal to form its shell. Ca(UCO,)., = CaCOg + HoO -f

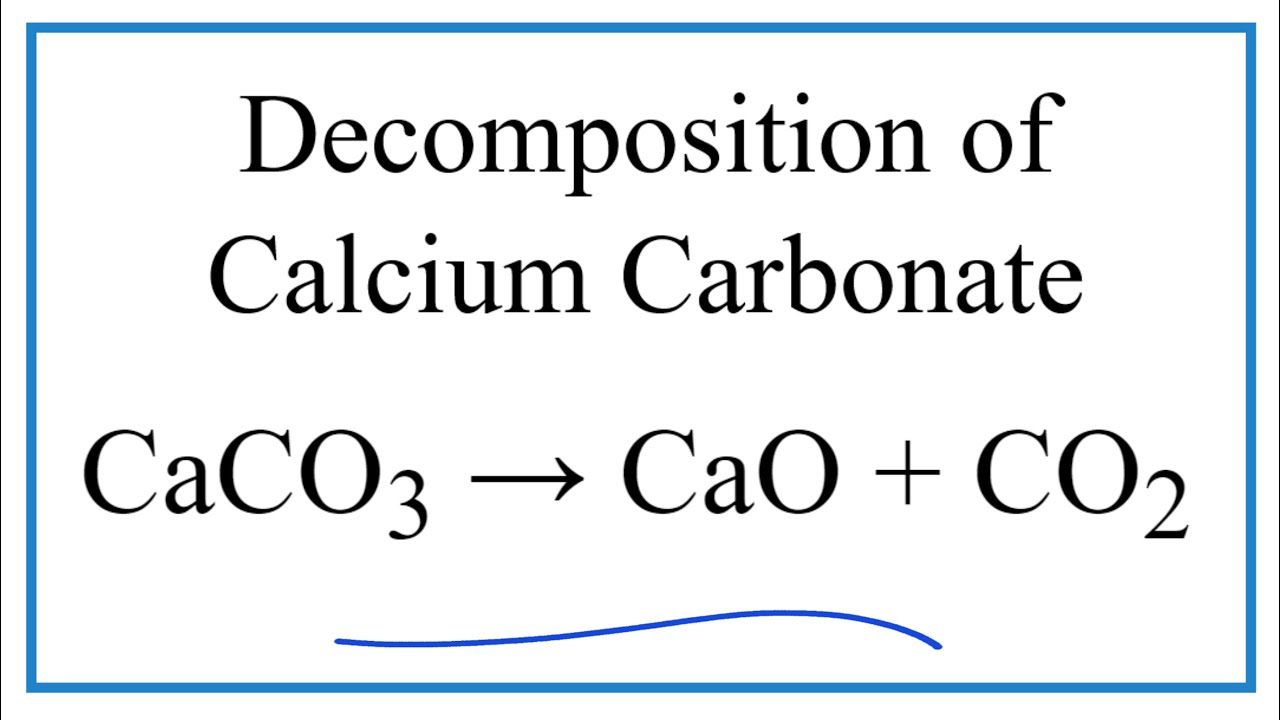
You can make chalk do something really weird just by heating it super hot. Chalk is made of calcium carbonate, and heating it causes the chalk to decompose into carbon dioxide gas and calcium oxide, ...

Draw the arrangement for heating of calcium carbonate and testing the gas evolved with burning match stick - Brainly.in